A brave little girl inspired a new ‘tumour paint’ to indicate to surgeons which tissue to remove.
In 2004 Dr Richard Ellenbogen spent almost 20 hours operating on a 17-year-old girl with a brain tumour. He ended up leaving a big piece of the tumour behind, mistaking it for normal brain tissue.
Less than a year after the surgery, the cancer hit back and the young girl died.
The week the girl died, Ellenbogen presented the case at his team’s weekly meeting at Seattle Children’s Hospital. “There’s got to be a way to take more of the tumour out and leave more of the normal brain intact.”
The nagging feeling that he could have taken out more tumour wouldn’t leave him.
Ellenbogen had faced a dilemma: if he had excised more, he would probably have removed more tumour but might also have removed normal brain tissue, with the risk that the girl would have been left severely disabled. Neurosurgeons have to be aggressive and sometimes push themselves to go further and deeper than they feel comfortable with, but they all operate under the adage “first, do no harm”.
Slight differences
The differences between cancerous and normal cells are often so minimal that they are extremely hard to tell apart. In the soft, gelatinous mass of the brain, Ellenbogen says tumour cells can feel like “fruit in [jelly]”, meaning that brain tumours are often slightly firmer and have a slightly more leathery texture.
Then again, sometimes the tumour can have the same texture as brain tissue. One could distinguish between the two through colour, but even then the difference can be slight. Ellenbogen spoke about one patient whose tumour was barely distinguishable from normal brain matter, except for a hint of yellow in the tumour cells.
There are several imaging technologies that help surgeons to see inside the body before they cut, many of which were developed to help diagnose cancer.
Ultrasound – bouncing high-frequency sound waves off structures within the body to see what they look like – was pioneered in 1942 by Karl Dussik, a neurologist at the University of Vienna, who attempted to locate brain tumours using essentially the same method bats use to navigate in darkness.
In October 1971, the first x-ray computed tomography scan (CT scan) helped to identify a frontal lobe tumour by producing cross-sectional images of the patient’s brain. CT scans are good at imaging dense materials such as blood and bone, so surgeons will use them, for example, if they are concerned about bleeding in the brain, in trauma where bones could be affected or in tumours that could involve bones.
Magnetic resonance imaging
One technology commonly used for imaging the inside of the body, especially soft tissues such as the brain, is magnetic resonance imaging (MRI).
Using a combination of radio waves and a very strong magnetic field, MRI provides information about where the tumour is and how it fits with other important structures in the body.
A more “real-time” version, called functional MRI, has also been used by surgeons to try to map out the safe and unsafe areas to operate on. Because these scans can show roughly which areas of the brain relating to brain function are affected, surgeons are able to give patients a better idea of what they might expect in recovery.
Increasingly, functional MRI is used in combination with intraoperative MRI, a technique developed about two decades ago in which scans are taken at intervals during surgery to check the progress of the procedure. This helps to reduce the risk of incomplete or damaging surgery, says Conor Mallucci, a consultant paediatric neurosurgeon at Liverpool’s Alder Hey Children’s Hospital in the United Kingdom. “There should be a 0% return-to-theatre rate for inadequate surgery,” he adds.
Potential of new technologies
But although all these technologies have improved things greatly, they are still not precise enough, especially with brain tumours. We are “still in the Middle Ages” when it comes to cancer surgery, says oncologist Dr Jim Olson.
“If you look at the rate of individuals that find out that they have bulky cancer left after their cancer surgery, it’s staggeringly high – for some cancers like brain cancer, it’s as high as 50%. For some very common cancers like breast cancer, it’s 30%,” he says.
What excites him is fluorescence imaging – a technology that literally lights up tumours so surgeons can see them. This doesn’t just have potential: it’s already proving to be effective in providing real-time image guidance to surgeons.
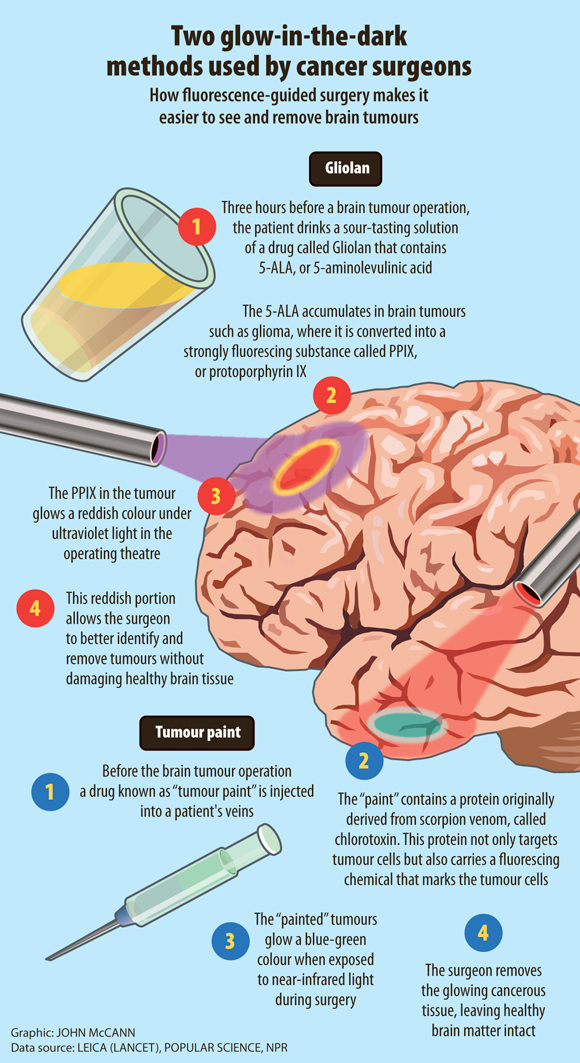
The dye 5-ALA, also called Gliolan, makes brain tumour cells glow red under ultraviolet light. The drug is swallowed by patients three to four hours before their surgery, to give it time to accumulate in their tumour cells.
It’s been approved for use in Europe since September 2007 but has yet to be approved in the United States, where it’s undergoing clinical trials. The US Food and Drug Administration has so far refused to approve it, citing that the original trial didn’t have overall survival as its primary outcome measure.
“I think it’s important for surgeons to have techniques they can use during surgery to identify where they should be removing disease [and], more importantly, to help distinguish areas that they need to leave behind and avoid,” says Dr Colin Watts of the department of neurosurgery at the University of Cambridge. Watts is leading a trial looking at whether 5-ALA could also act as a delivery device for the chemotherapy drug Carmustine, which could be implanted into the cavity of the removed tumour to kill leftover tumour cells after surgery.
Fluorescence imaging could allow surgeons to identify tissue that needs to be cut out, such as tumours, and tissue that needs to be avoided, such as blood vessels and nerves. The visual can also help to determine whether a tumour can be operated on and, if necessary, what type of follow-up therapy – be it chemotherapy or radiation therapy – would be needed. This added insight allows surgeons and their patients to make more informed decisions about their treatment.
The past few years have seen an explosion of proof-of-concept clinical trials in the field of fluorescent image-guided surgery, but one method might be even better than 5-ALA. It’s called tumour paint.

Violet’s legacy
Jim Olson remembers being ridiculed. It was 1989 and he was defending his PhD thesis, and the University of Michigan professors asked what his next goal would be. “If we can bring radioactivity into these tumours for PET scanning, I would love to find a way to bring light into the cancer so that surgeons can see it while they’re operating,” Olson told them. The professors chuckled. “Okay, Buck Rogers,” one of them heckled, “but what are you really going to do?”
Positron emission tomography (PET) scanning uses radioactive tracers to find cancerous cells. Unlike ultrasound or CT scanning, it detects differences in tissue based not on its structure but on a measure of some metabolic change – the uptake of sugar, for instance. This can distinguish a live tumour from a treated tumour or a tumour that’s dying. However, surgeons seldom use the technique because it is considered too blunt and unable to give enough precise detail. Moreover, it’s not suitable for children because exposing their developing bodies and brains to radiation could lead to other forms of cancer later in life.
A more accurate, radiation-free method that directly targeted cancer cells would be revolutionary, not least for childhood cancer patients. So when Ellenbogen spoke at that Seattle Children’s Hospital meeting in 2004, the eyes of Olson, sitting across the table, lit up.
Cancer in children
Cancer can occur at any age. Among children under the age of 19, tumours in the brain and central nervous system are the most common cancers.
Every year, in the US alone, about one in 285 children – nearly 16 000 in total – will be diagnosed with cancer before their 20th birthday, and more than a quarter of them will have been diagnosed with a brain tumour.
In most childhood brain tumours, a complete removal improves the overall survival rate.
Olson has diagnosed babies with cancer at one day old, and some even before birth.
The toughest thing about being an oncologist, Olson says, is telling parents – not to mention the child – that a cancer has come back. “It’s one thing to say: ‘Your child has cancer and this is the plan, and this is what to expect,’” he says. “It’s another thing to begin a conversation that says: ‘Your child has recurrent cancer, and it’s extraordinarily rare for anybody to survive this.’ You’re preparing for death and preparing for the miracle at the same time.”
No hope for Violet
There was no miracle for 11-year-old Violet O’Dell. She had a large, rare kind of brain tumour. The cancer had woven itself in between the normal nerves of the brainstem – the lowest part of the brain, which connects the brain to the spinal cord and is involved in basic functions such as breathing, heartbeat and blood pressure, and some reflex actions such as swallowing. It was impossible to remove the cancer without killing her.
The day she was diagnosed was terrifying, Violet’s mother Jess tells me, but at the same time it was a kind of relief: there was a sense of: “Okay, we know now.” Violet had been falling asleep in class, the beginning of an odd set of behaviour changes. “For months we had been having problems with her acting odd, belligerent, slurring and stumbling … I would constantly be telling people: ‘I don’t know where she went. Whoever this is, this isn’t Violet.’” After six months of investigation, an MRI scan found the tumour. The diagnosis was given at the Seattle Children’s Hospital by Olson.
Olson recalls the conversation he had with Violet when she came to visit his lab. A few days earlier she had seen her mother’s driving licence with a heart symbol on it, which indicated that Jess was an organ donor. Violet asked if she could donate her organs too, but Jess wasn’t sure because of her cancer.
Hope in research
The then 10-year-old Violet understood that the kind of cancer she had was inoperable, and she knew she was going to die. And after she met Olson’s team, who told her about their work, she wanted to know what was being done to research her type of tumour.
It was a tough question for a doctor to answer. Olson told her that it was difficult because researchers didn’t have material – tumours – to work with. The little girl responded: “When I die, I want you to do an autopsy, take my tumour and put it into mice. So that you can study my cancer and help other kids that get it in the future.”
Violet died a year later at her grandparents’ home, beside her family and pets: a puppy, two Labradors and a cat. A few weeks after her death, Olson honoured her by naming his new study Project Violet. He hoped it would revolutionise cancer surgery with a drug from an unusual source.
Scorpions to the rescue
Professor Harald Sontheimer, then at the University of Alabama, was never really interested in cancer. If it wasn’t derived from his favourite cells, glial cells – a type of cell in the nervous system from which most types of brain tumour develop – he probably wouldn’t be studying it.
Initially, it was thought that glial cells simply kept everything glued together (the term “glia” stems from the Greek word meaning “glue”), but about 30 years ago scientists found out that they do much more. In addition to surrounding neurons and holding them in place, glial cells have three other main functions: supplying nutrients and oxygen to neurons, insulating neurons from one another, and killing pathogens and removing dead neurons.
They also help to maintain the blood–brain barrier, which filters substances before they reach the brain, and play a part in the regulation, repair and regrowth of tissue after damage.
There are about 130 different types of brain tumours; the most common type, which is known as a glioma, develops from glial cells. In 1995 Sontheimer and his research team were interested in glial cell-derived tumours.
The team had identified that chloride channels – key mechanisms in cells – were somehow involved in tumours invading brain tissue. They were experimenting with substances that would block these chloride channels. The most effective by far was chlorotoxin, a substance isolated from the venom of the deathstalker scorpion.
Venom trials
It is one of the most deadly scorpions in the world. Found in arid and hyper-arid regions of North Africa and the Middle East, the Palestine yellow scorpion is about 10cm long but can kill creatures a thousand times its size.
In humans, the scorpion’s sting can cause excruciating pain, convulsions, paralysis and potentially even death owing to heart and respiratory failure, but most affected humans will just suffer extreme pain in the region of the sting, along with drowsiness, fatigue, splitting headaches and joint pain. These symptoms are caused by the scorpion’s powerful venom, which is a mixture of neurotoxins – poisons that act on the nervous system – including one known as chlorotoxin.
Sontheimer’s team took a human brain tumour isolated from a patient and introduced it into the brain of a mouse, then injected synthetic chlorotoxin they had created in the laboratory. What they found was remarkable and unexpected. The synthetic chlorotoxin accumulated in tumours – and only tumours – leaving normal, healthy cells untouched.
It also showed an ability to pass into the brain, unobstructed by the blood–brain barrier.
As Sontheimer explained in a later paper: “They are opening the blood–brain barrier specifically at the point where they’re moving along the blood vessels. They’re essentially ensheathing the blood vessels, and as they do that, they’re gradually breaking down this barrier so that, at the site where they’re invading, there is active penetration of this molecule into the brain.”
Illuminating cancer
In 2004 Ellenbogen sent one of his neurosurgery residents, Patrick Gabikian, to Olson’s lab at Seattle’s Fred Hutchinson Cancer Research Centre to complete a year of research. There, Gabikian would begin looking for compounds that Ellenbogen and Olson could use to illuminate cancerous cells. One day Gabikian came across a compound that Olson saw as a viable option to experiment with: chlorotoxin.
In 2007 Olson, Ellenbogen, Gabikian and their team wrote an article detailing how they had isolated chlorotoxin from the deathstalker scorpion venom and attached a fluorescent molecule, producing a substance that would “light up” tumour cells. Like Sontheimer, Olson’s lab uses a synthetic version of the chlorotoxin protein, but the added fluorescent molecule acts like a flashlight – turned on only when it binds to its target.
That target is thought to be a complex containing the protein annexin A2. In normal noncancerous cells, it lies inside the cell, but in cancerous tissue cells it appears to be turned to the outside of the cell’s surface. When chlorotoxin binds, the complex gets moved to the inside of the cell, carrying the flashlight molecule with it. And when that happens, cancerous cells emit a light that can be readily detected by a variety of devices.
By making even the smallest pockets of cancerous cells visible in real time during surgery, this “tumour paint” – BLZ-100 – can help to determine the precise location and size of the primary tumour and its satellites, which are smaller areas of tumour located nearby. This, Olson decided, was a concept worthy of Project Violet. He had the technology. Now what he needed was funding.
Crowdfunding for science
It can take years for government grants to come through, if your application is even successful in the first place. Olson decided a long time ago that applying for grants was just delaying his research. Today, his main financial backing comes from the families, individuals and family-run foundations that have heard about his research. He tells of one father who brought his daughter to the clinic for a flu shot, heard about Olson’s work and dropped off a cheque for $100 000.
“Having to say ‘no’ to Jim is like punching yourself in the face,” says Nicole Pratapas, philanthropic gifts adviser at the Fred Hutchinson Cancer Research Centre.
She meets him every Tuesday morning at his office. “The way that he feels about his patients, and how we do things, it means a lot to us.”
In the past two years alone, Olson has raised $6 million through private funding.
That, however, is earmarked for staff costs and the multitude of other research projects he has in the works. When tumour paint started to seem viable, he knew he had a winning idea but had no spare budget to start developing it. What he did have was a short film.
Attention
Bringing Light is a three-minute documentary by Bert Klasey, Chris Baron and James Allen Smith. The film was one of 20 finalists at the 2013 Focus Forward Filmmaker competition and won the Audience Favourite award.
It came about when producer Klasey was looking for ideas and his wife remembered a presentation that had stuck with her since she’d seen it at a conference, eight years previously. It was Olson, talking about what would become tumour paint.
The film presented Olson with an opportunity. He thought he could use it to harness two of the most powerful tools of the modern entrepreneur: goodwill and crowdfunding. As it happens, just a few blocks away from him were the offices of the world’s biggest internet retailer, Amazon.
He sent them a link to Bringing Light and invited the Amazon team to visit his lab, meet the scientists over beer and pizza, and hear more. Olson expected that about 20 people would turn up. More than 70 did – ever the optimist, Olson had catered for 80, “just in case”. In just a few weeks, 25 of them had formed a volunteer project team that would give up every Monday evening for the next year to design Project Violet’s website and put the word out on Facebook and Twitter.
Project Violet is a citizen science project kick-started via an online fundraising campaign, originally based around a drug adoption programme – for a $100 donation anyone could “adopt” a potential drug target, which would go toward its research.
This has since been removed because, according to Olson, people found it daunting to adopt a drug, wondering if they were smart enough to choose the “right” one. In less than three years, Olson and his team have raised $5 million through the Project Violet website and related events, combined with individual donations.
Tumour paint
Cameron W Brennan, a neurosurgeon at the Memorial Sloan Kettering Cancer Centre in New York, views this without cynicism. In fact, he applauds the press coverage: “Olson has to focus fundraising and to focus research on things that will affect practice.” And Olson has proven before that he is really good at that.
It’s taken nearly 25 years, but Olson is on track to make his and Ellenbogen’s vision a reality. During preclinical trials with mice and dogs that took place between 2005 and 2011, tumour paint proved to be more than 5 000 times more “sensitive” than MRI. It could highlight extremely small numbers of cancer cells – as few as 200 cells – whereas MRI has a lower limit of at least half a million.
Unlike 5-ALA, tumour paint can be used in real time during an operation, and Olson points out that it also crosses the blood–brain barrier, attaches specifically to cancer cells and is internalised. The 5-ALA dye, by contrast, doesn’t bind to cancer cells and can only be used for high-grade brain tumours that are highly malignant and adept at invading nearby brain tissue. Tumour paint has the potential to target lower-grade tumours as well – which is important, says Olson, “because low-grade gliomas that are incompletely removed often progress to high-grade glioblastomas over the following decade”.
On May 25 2015, the first clinical trial of tumour paint began at Seattle Children’s Hospital, which has the largest paediatric brain tumour centre in the northwest of the US.
The trial is run by Blaze Bioscience Inc, and in phase one up to 27 people who have been diagnosed with a brain tumour – from infants to young adults under the age of 30 – will have their operations performed using the drug.
“I think lots of other people are trying to find out the difference between brain cancer and normal tissue, but they don’t know what to do with the information when they get it,” Olson says. “They publish their paper and then they move on to the next thing.”
He hopes tumour paint will be different. If it is, it will go some way towards living up to Project Violet’s name.
“All these buildings and universities get named after wealthy people that donate money,” Olson says. “I wanted something really beautiful for the world to be named after this little girl.”
This story first appeared on Mosaic and is republished here under a Creative Commons licence. The original article can be found at mosaicscience.com/story/tumour-paint-cancer-surgery

